Fuel can be produced by combining the hydrogen produced from solar energy with greenhouse gases emitted by humans. A method to convert CO2 into energy has been proposed by Dr. FUJISHIMA Akira, the discoverer of photocatalysis.
It is not widely known that the glass pyramid in the courtyard of the Louvre Museum in Paris has a transparent coating that exhibits an antifouling effect upon exposure to natural light. Dr. FUJISHIMA Akira, a Japanese researcher, pioneered the discovery of that photocatalysis reaction, which involves the principle of breaking down dirt without using any energy or incurring any cost. Although photocatalysis has been widely developed and put into practical use in the areas of antifouling and antifogging, research on artificial photosynthesis—the process of extracting hydrogen through photocatalysis—has also been garnering significant attention in recent years as a technology with the potential to contribute to a decarbonized society.
It was in 1967, while he was at a graduate school under the supervision of the late Dr. HONDA Kenichi, that Dr. Fujishima, alongside his supervisor, discovered the photocatalysis reaction. This phenomenon produces hydrogen and oxygen when titanium oxide is exposed to light under water, in effect reproducing the redox (or oxidation-reduction) reaction similar to that of photosynthesis in plants. So excited was Dr. Fujishima with his discovery—the knowledge that oxygen could be extracted simply through exposure to light—that he was unable to sleep for some time.
Dr. Fujishima’s paper, written jointly with Dr. Honda, was published in the journal Nature in 1972, following which the photocatalysis reaction became known as the Honda-Fujishima effect. Ever since the oil crisis of the 1970s, the process of being able to extract hydrogen through photocatalysis has been greatly anticipated as a source of alternative energy to replace oil, and researchers around the world have been working on an efficient means to extract hydrogen via artificial photosynthesis using that principle.

Photocatalysis acts as an antifouling and antifogging effect simply by the irradiation of light. It has also been used in the glass covering the pyramid-shaped entrance of the Louvre Museum. The glass maintains its transparent beauty by decomposing dirt.

Fujishima (left) conducting an experiment with Dr. Honda in 1967. Initially, nobody believed the pair’s findings, namely, that water could be broken down with light energy alone.
With the United Nations Sustainable Development Goals (SDGs) aiming to reduce greenhouse gases, research on artificial photosynthesis has been increasingly gaining pace, though many challenges still remain. Dr. Fujishima says, “To achieve the practical applications of hydrogen production using artificial photosynthesis, the high efficiency of hydrogen extraction is, of course, the basic key factor. However, the other key factors are whether we can find a catalyst that satisfies the remaining various conditions; this includes whether the materials used as catalysts can be easily obtained, whether a large surface area photocatalyst can be manufactured, and whether any harmful substances are contained in the material. We are waiting for a breakthrough for those things in the future.”
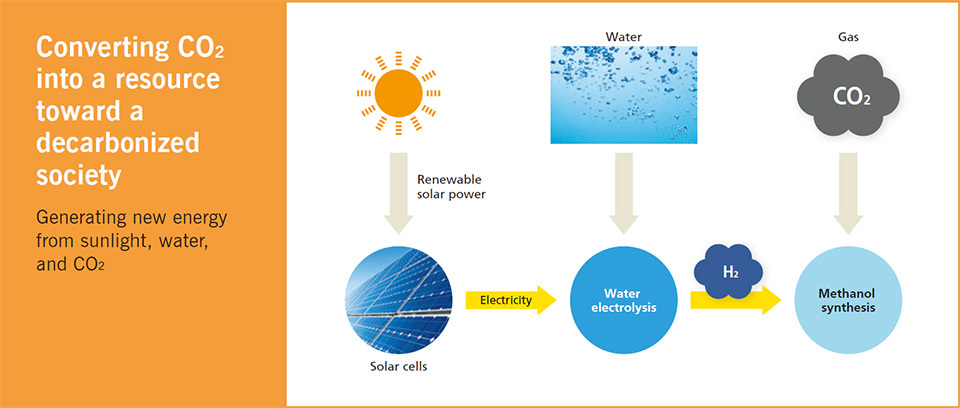
Along with the shift to replace fossil fuels with renewable energies such as hydrogen, another important measure to achieving a decarbonized society is carbon recycling, effectively using CO2 as a resource. In consideration of that viewpoint, Dr. Fujishima has proposed the following method; first, extract hydrogen through water electrolysis using the electricity produced from highly efficient solar cells. Next, combine the extracted hydrogen with the CO2 emitted from power plants and factories to produce methanol, which can be used as an energy source. If this process is realized, gases containing carbon, such as CO2, will no longer contribute to the greenhouse effect. Rather, those gases will become “resources” to replace oil and natural gas.
Dr. Fujishima currently heads the Photocatalysis International Research Center at the Tokyo University of Science, where he is leading research on further progress of photocatalysis in the fields of the environment and energy. “I think it’s important for science to contribute to the world,” he says. Based on his discovery, a decarbonized society, where even CO2 is used as a resource, will soon be realized.