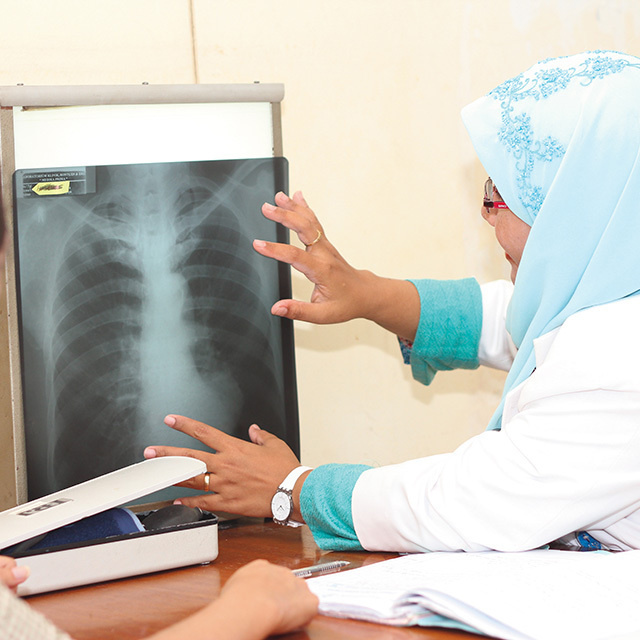
As of mid-May 2020, more than 4.4 million people worldwide have been infected with the novel coronavirus and have come down with COVID-19, over 300,000 of whom have lost their lives. With the race on to develop therapeutics, one scientist is fighting to save lives.
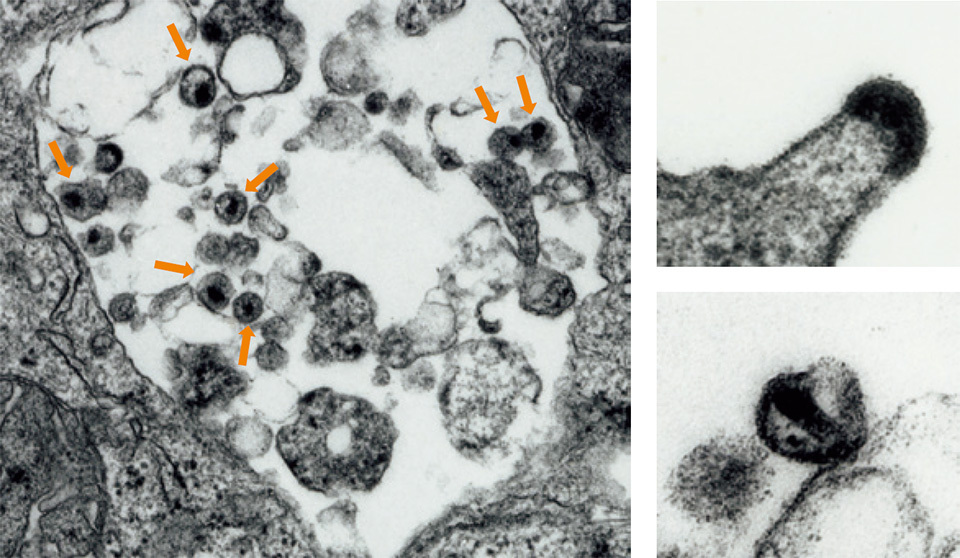
Top left: When HIV infects a lymphocyte, a type of white blood cell that plays a major role in the body’s defense, copious numbers of the virus (arrows) emerge, destroying the cells.
Top right: When HIV emerges through the membrane of the infected cell, each virus leaves the cells by “budding”.
Lower right: After HIV emerges from human cells, the virus matures to have a dark “core” and becomes infectious.
“We certainly will be able to develop therapeutics for COVID-19.” The man behind that powerful statement is Dr. MITSUYA Hiroaki, a scientist who was a primary contributor to the discovery of the first three medications ever for AIDS, the acquired immune deficiency syndrome that once terrorized the world as a death sentence to those it afflicted. He later went on to help develop the fourth therapeutic treatment for AIDS.
The human immunodeficiency virus, or HIV, is the virus that causes AIDS. HIV enters the immune cells of the human body, where it hides inside their genes and replicates within the cells before eventually destroying those critical cells, thus rendering the immune system powerless. The result is that AIDS patients later become infected with other viruses and bacteria or develop cancers, which they are unable to fight against, eventually leading to their death.
Azidothymidine (AZT), the first-ever anti-HIV medication, which Dr. Mitsuya was directly involved in developing, attacks the reverse transcriptase of HIV. This enzyme regulates the replication process that HIV must undergo to multiply. By halting the reproduction of the virus’s genes, AZT stops the virus from replicating. Didanosine (ddI) and zalcitabine (ddC), the second and third drugs—the development process in which Dr. Mitsuya played an indispensable role—likewise inhibit the mechanism behind HIV’s propagation. Since human cells do not possess reverse transcriptase, attacking them does not prevent our body’s cells from multiplying or functioning: it only stops the virus from multiplying.
Darunavir, the fourth anti-HIV medication, for which Dr. Mitsuya also played a central role in developing, comes from a “second generation” of drugs. This drug targets the protease, another kind of enzyme that breaks down proteins by cutting premature viral proteins like a saw or a pair of scissors. When HIV infects human cells, it takes over their protein synthesis function to begin making HIV proteins. To do so, HIV first makes the cell produce large virus proteins. It then uses its own protease to precisely cut those large virus proteins into small, mature, and functional proteins. Next, those parts are assembled into virus particles. HIV protease appears and functions in a way different from that of the proteases in human cells. Therefore, attacking HIV protease does not interfere much with the functions of human cells, all the while impeding the formation of virus particles. Such protease inhibitors against HIV have proven to be a powerful deterrent to HIV reproduction as well.
It has been 33 years since the first anti-HIV medication, Dr. Mitsuya’s AZT, was approved. After that pioneering drug, many more powerful medications have been created with fewer side effects and simpler administration. Thanks to that drug development, AIDS has gone from a disease that at its peak took 1.7 million lives a year and was fatal to nearly 90% of patients within two years[1], to a controllable chronic disease, allowing people living with HIV to pursue careers, raise families and children, and lead lives much like those who do not have the virus. Treatment can also prevent people living with HIV from infecting other people.

Dr. MITSUYA Hiroaki currently serves as director of the National Center for Global Health and Medicine (NCGM), located in Tokyo, and head of the HIV and AIDS Malignancy Branch’s Experimental Retrovirology Section at the National Cancer Institute, part of the National Institutes of Health (NIH) in the United States.
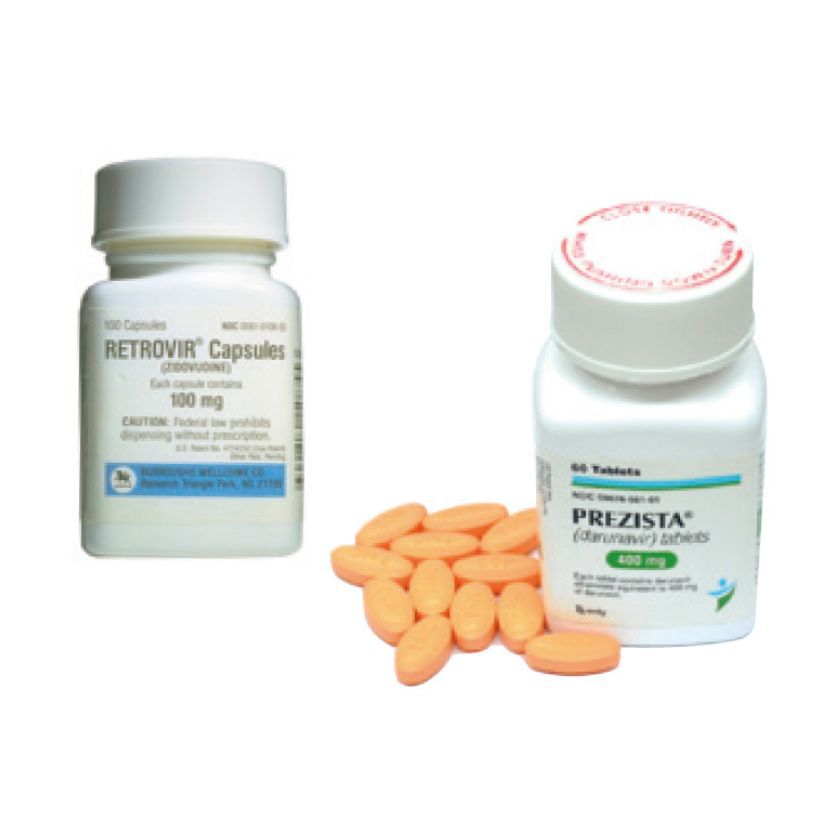
AZT (left), the first anti-HIV medication ever developed in the world. The second-generation drug darunavir (right), is the world’s first registered anti-HIV medication available for use by developing countries with patent royalties waived.
Dr. Mitsuya is now working to develop therapeutics for COVID-19 in a partnership with the National Center for Global Health and Medicine (NCGM) in Japan, as well as with scientists and the National Institutes of Health (NIH) in the United States. SARS-CoV-2, also known as the novel coronavirus, is the pathogen that causes COVID-19. To develop therapeutics for SARS-CoV-2, scientists target the virus much in the same way that they have done with HIV. An examination of infections around the world strongly suggests that it may not be a seasonal virus, which means it could continue to pose an immense threat to humans throughout the year. It is as yet unknown whether people who develop antibodies against SARS-CoV-2 gain long-term immunity. There is still much to learn about the virus, but according to Dr. Mitsuya, “there are many lessons we can learn from the development of drugs to treat AIDS”, which can be applied to new therapeutics for combating SARS-CoV-2.
Dr. Mitsuya sees three targets in attacking the virus. The first is when SARS-CoV-2 enters a human cell. The second is the virus’s unique RNA polymerase that mediates the reproduction of SARS-CoV-2 genes. The third is the protease of SARS-CoV-2, in particular, the main protease that plays a primary role.
The main protease has a different structure and function from those of human proteases that break down proteins in human cells. According to Dr. Mitsuya, researchers have already obtained chemical compounds in test tubes that exhibit potent effects against SARS-CoV-2. He claims that he has also already identified multiple novel chemical compounds that completely block SARS-CoV-2 from infecting cultured cells, without having any major impact on human cells.
Dr. Mitsuya goes on, “We’ve also administered some of them to mice, and they remained apparently unaffected. We still need to examine them in clinical trials, but I have high hopes that several of such chemical compounds will not be acutely toxic and will be efficacious against the virus.” However, when drugs that have tested as beneficial to animals are administered to patients in clinical trials, they do not always have the expected effect. In many cases, extreme side effects are observed.
Cautioning that it is too soon to judge, Dr. Mitsuya continues, “When we developed AZT, the first anti-HIV medication, we broke records in that we won approval in only 25 months after discovering its effect against HIV. The approval process had never moved with such speed before. Now we want to do that in a year.” When AZT was first reported, Dr. Mitsuya said he already had ddI and ddC on his hands. He adds, “I have the same situation now.”
Looking back, Dr. Mitsuya concludes that AZT was “a tough medicine” because of its strong side effects. “However, it surely helped save lives. If you can help save lives, then you can come up with better and better drugs to make patients healthy again.” Once a body is infected with HIV, the virus embeds itself in critical cells’ genetic codes, which means that the virus can seemingly never be completely removed. SARS-CoV-2, though, does not enter the genes. That means that we “can no doubt eradicate it.” The scientist’s words carry great weight. (Interview conducted on April 24, 2020.)
[1] UNAIDS DATA 2019
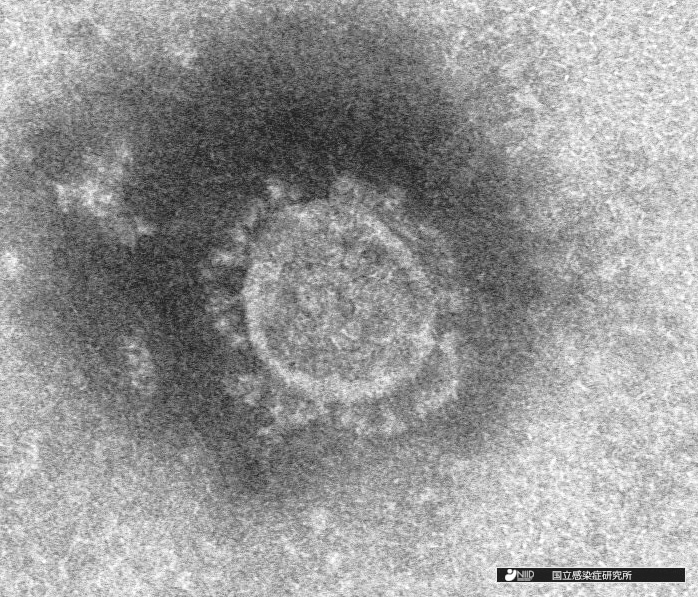
Electron-microscope image of the novel coronavirus isolated at the National Institute of Infectious Diseases (NIID) in Japan.
Japan’s National Center for Global Health and Medicine (NCGM), under Dr. Mitsuya’s direction, is dedicating all its strength to the development of new drugs.
Latest Report on Mitsuya’s Drug Development Research in Response to the COVID-19 Pandemic
GRL, which targets the essential enzyme (main protease: Mpro) of SARS-CoV-2, completely protects VeroE6 cells from SARS-CoV-2 infection.
When VeroE6 cells, which are susceptible to SARS-CoV-2 infection, were cultured alone, they stain in red (showing healthy act in filaments of normal cells) and in blue (showing the nuclei of the cells). When VeroE6 cells were exposed to SARS-CoV-2, the cells additionally stain in green (showing the presence of virus antigens). However, when VeroE6 cells were exposed to SARS-CoV-2 and cultured in the presence of a novel small chemical, named GRL, the cells were completely protected by GRL from SARS-CoV-2 infection and the cells do not stain in green. The staining method is called immunocytochemistry.
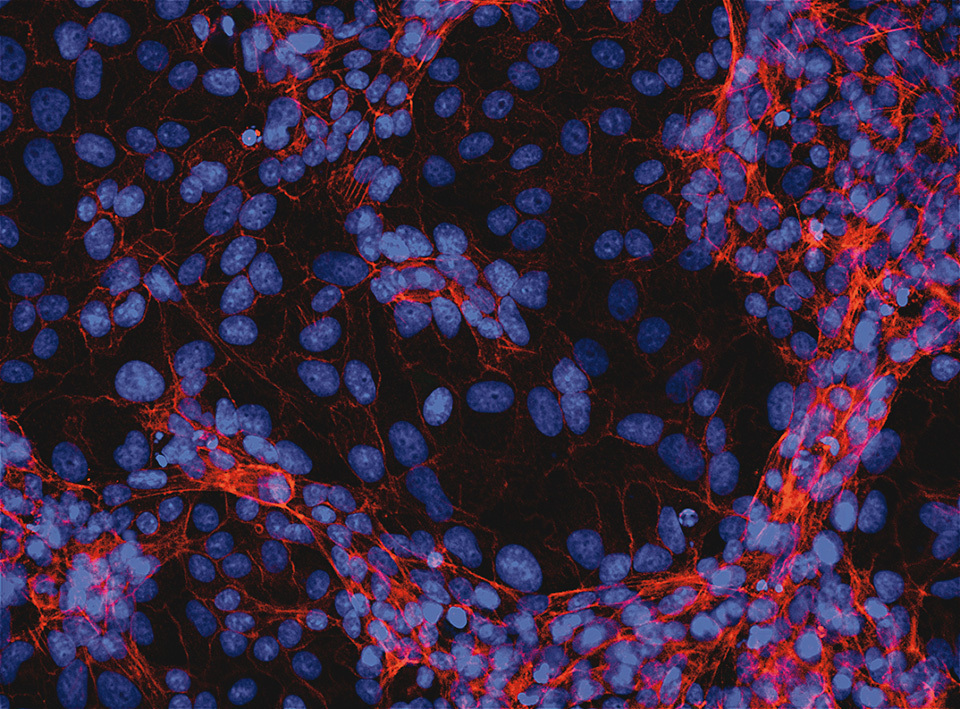
VeroE6 cells alone
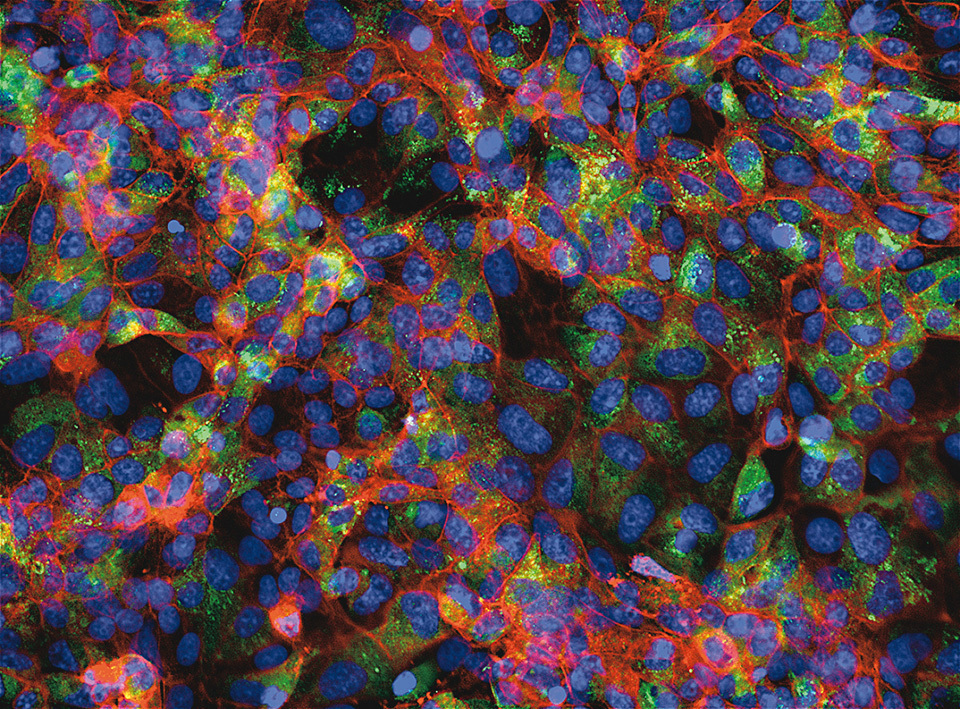
VeroE6 cells exposed to SARS-CoV-2
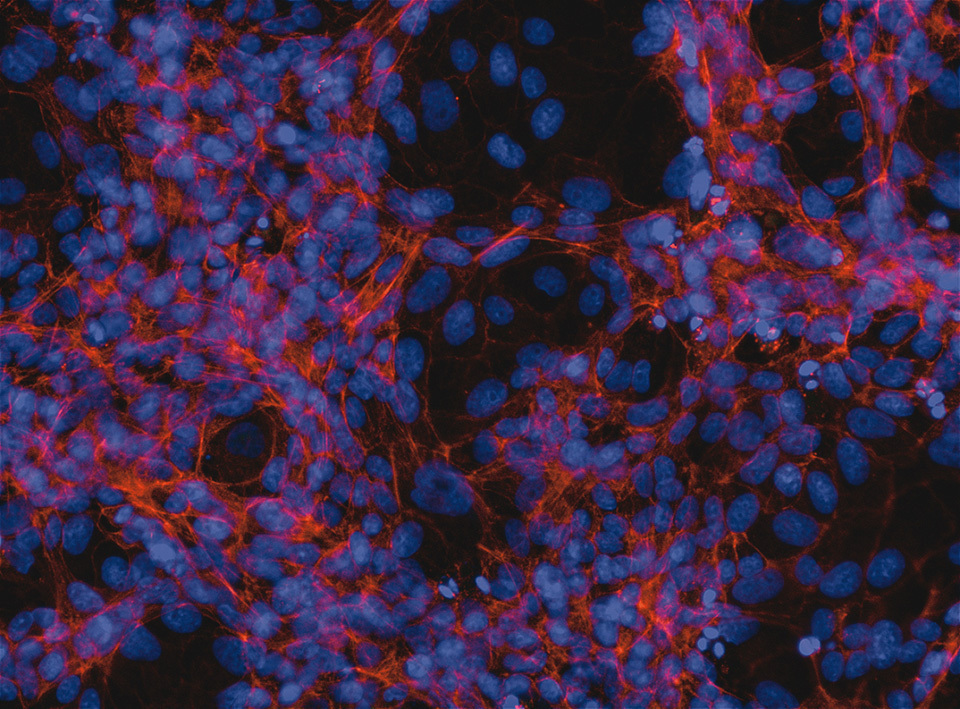
VeroE6 cells exposed to SARS-CoV-2 but protected by GRL