Fuel can be produced by combining the hydrogen produced from solar energy with greenhouse gases emitted by humans. A method to convert carbon into energy was proposed by Dr. Akira Fujishima, the discoverer of photocatalysis.

Photocatalysis acts as an antifouling and antifogging effect simply by the irradiation of light. It has also been used in the glass covering the pyramid-shaped entrance of the Louvre Museum. The glass maintains its transparent beauty by decomposing dirt.

Dr. Akira Fujishima is the director of the Photocatalysis International Research Center at the Tokyo University of Science. He discovered photocatalysis while enrolled at the University of Tokyo Graduate School. That discovery was later called the Honda–Fujishima effect, with research on artificial photosynthesis then being initiated around the world.
The phenomenon of photocatalysis, announced in 1967 by Dr. Fujishima along with Dr. Kenichi Honda, is able to produce a variety of effects using only light, and without using any electricity. Its applications have since expanded to supplying antifouling and antifogging effects, as well as oxidation-reduction, among many others. Dr. Fujishima’s research regarding artificial photosynthesis, which was concerned with extracting hydrogen from water and using it as energy, has gained a particularly large amount of attention.
Dr. Fujishima says, “To achieve the practical application of hydrogen production using photosynthesis, the high efficiency of hydrogen extraction is, of course, the basic key factor. However, the other key factors are whether we can find a catalyst that satisfies the remaining various conditions; this includes whether the materials used as catalysts can be easily obtained, whether a large surface area photocatalyst can be manufactured, and whether any harmful substances are contained in the material. We are waiting for a breakthrough for those things in the future.”
The reduction of greenhouse gases is also a prerequisite for achieving Sustainable Development Goals (SDGs). As Prime Minister Shinzo Abe stated at the World Economic Forum this year, an existing technology of methanation to remove CO 2 is getting attention anew by creating resources through the combination of CO 2 and hydrogen, in efforts toward the realization of a decarbonized society.
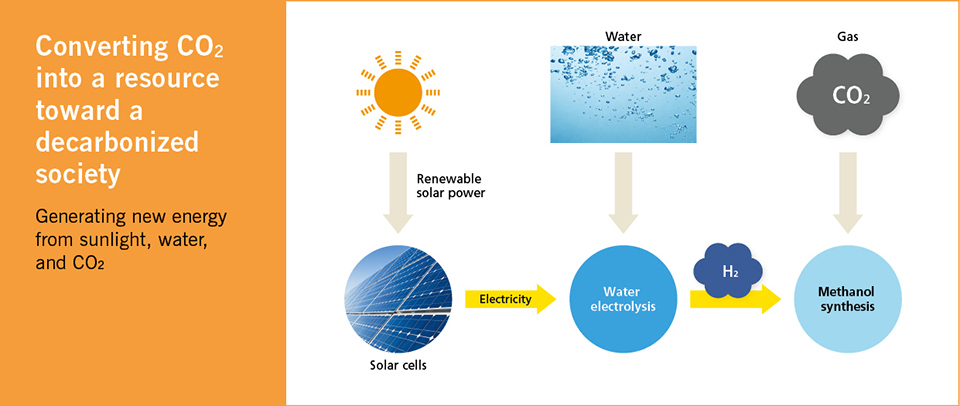
In consideration of that viewpoint, Dr. Fujishima has proposed the following method; first, extract hydrogen through water electrolysis using the electricity produced from highly efficient solar cells. Next, combine the extracted hydrogen with the CO2 emitted from power plants and factories. Finally, synthesize methane gas, which can be used as an energy source. If this process is realized, gases containing carbon, such as CO2, will no longer contribute to the greenhouse effect. Rather, those gases will become “resources ” to replace oil and natural gas. This mechanism is known as carbon recycling. Dr. Fujishima added, “The production of methane is a methodology to contribute to both climate change and the resource issue. At present, we must overcome a number of big hurdles, and I am also actively conducting research on them. ”
The innovation of efficient carbon recycling will surely lead to the resolution of global issues.

Japanese high school students, Kizu (left) and Matsumoto, discovered that the ingredients contained in used tea leaves can enhance the photoreduction of iron ions.

Used Tea Leaves Found to Enhance Photoreduction
Similar to Dr. Fujishima, a scientist who opened the door to a new world of science more than half a century ago, several young scientists are working every day making new observations that will lead to the future. Two such young scientists are Hisato Kizu and Hayato Matsumoto from the Shizuoka Kita High School Science Club, who devised a method to effectively produce hydrogen from water by utilizing used tea leaves.
The two young scientists initially demonstrated that when iron ions are used as catalysts, with the oxidation of iron ions, water electrolysis occurs at a voltage lower than that which is found under normal conditions. Furthermore, inspired by the fact that iron ions and green tea are used for dyeing fabrics, they repeated the experiments considering that the polyphenol contained in tea may be effective in the reduction of oxidized iron ions. Consequently, they observed that photoreduction is promoted by the used tea leaves. These scientists also participated in and presented their research at the Stockholm Junior Water Prize held in Sweden in August.
Shizuoka is known as the prefecture which produces the most tea in Japan. Although Japanese tea is said to contain a high polyphenol content and thus is good for health, used tea leaves are generally discarded. However, the leaves would not be discarded if they were utilized as a catalyst that produces hydrogen as an energy source in a low-cost and eco-friendly way. Such a day may come in the future. “We want to become useful people for the world,” the two students said with a smile.