In 2016, the Nobel Prize in Physiology or Medicine was awarded to Dr. Yoshinori Ohsumi of the Tokyo Institute of Technology for elucidating the mechanisms underlying autophagy. Autophagy (a term originating from the Greek words auto -, meaning “self,” and phagein , meaning “to eat”) is a vital function in which intracellular protein is broken down to be reused for generating the amino acids needed to sustain life. This function is also known as the “intracellular recycling system.”
Dr. Ohsumi was the world’s first to observe the autophagy process through an optical microscope in 1988. The professor was studying yeast cells, which are commonly used in cell research, and was focused on a particular organelle—the vacuole. He was working to elucidate the mechanism of its degradative function in yeasts, a function that was not clear at the time. As a result, he confirmed that cytoplasmic components such as proteins are taken in by the vacuole. Dr. Ohsumi recalls, “It was an awe-inspiring sight that I could have kept watching for hours. At that point, I didn’t grasp the full significance of what I was watching, but I knew I’d made a very important discovery. For me, that was the breakthrough moment that I would look back on whenever I felt discouraged by not yet being able to see the true nature of what I was studying.”
Dr. Ohsumi participated in the Nobel Prize Award Ceremony held in Stockholm on December 10, 2016. The professor received his medal for Physiology or Medicine from His Majesty King Carl XVI Gustaf of Sweden.
Dr. Ohsumi and members of the Tokyo Institute of Technology’s Ohsumi Laboratory. The laboratory has a friendly atmosphere, and the members are drawn by the professor’s passion for research and his unpretentious manner.
From 1996, Dr. Ohsumi worked together with laboratory members to determine that autophagy was a process not just limited to yeast, but was a shared mechanism in all animal and plant organisms. “Although my study was focused on yeast, the laboratory at the time accommodated several outstanding researchers who were studying the cells of plants and animals,” he recalls. “These members joined in my research to form an ideal team. I believe that the time spent drinking with them and discussing our research contributed to the rapid progress of my study.”
Today, autophagy research is being intensely pursued all over the world. It has been discovered that some illnesses such as Parkinson’s disease are triggered by the accumulation of abnormal proteins resulting from disrupted autophagy in the nerve cells of the brain. Elucidating the basic mechanisms of autophagy may possibly lead to discovering the causes of illnesses as well as the development of cures, and is expected to contribute towards defining the mechanisms of aging and metabolism.
Regarding his approach as a researcher, Dr. Ohsumi says, “The thought that drives me to study the subjects that I want to know has always been, ‘Do what no one else is doing.’ My mission is to shed light on the fundamental functionality of the cell. I want to keep moving forward with my studies to help solve the biological mysteries of life.”
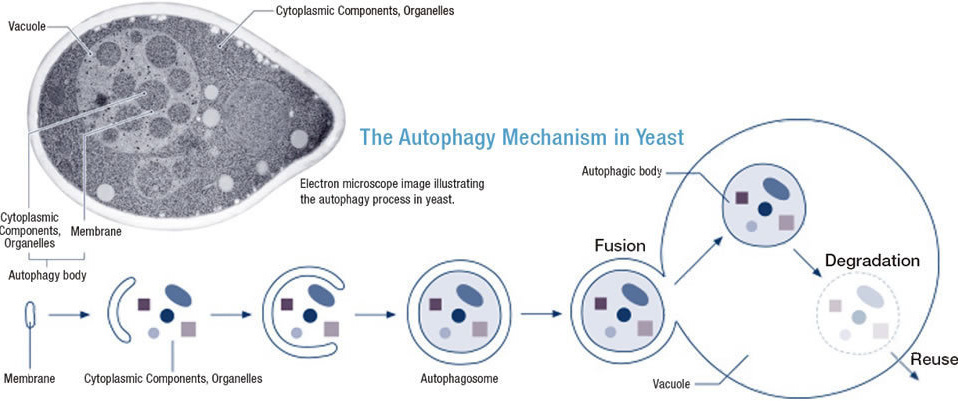
In yeast autophagy, a membrane is initially formed within the cell. This membrane grows to completely wrap around proteins and other cytoplasmic components that are targeted for degradation. The resulting double-layer membrane is called an “autophagosome,” the outer membrane of which can then fuse with the vacuole membrane. The inner membrane and cargo of the autophagosome are thereby released into the vacuole, where the structure is referred to as an “autophagic body.” The membrane and contents of the autophagic body are finally broken down by degradative enzymes within the vacuole. The raw components derived from this degradation can then be returned to the cytoplasm for reuse.